INHI-SIAL


« Synthesis of sulfur analogues of DANA, potential inhibitors of human neuraminidase hNEU1. »
Co-financed by the Grand-Est Region and the ERDF
Amount of aid granted: 48 140 €
Duration: 1 year (September 2021 - August 2022)
Project leaders : Dr. Murielle Muzard, Pr. Richard Plantier-Royon
During aging, the degradation of elastic fibers, mainly composed of elastin, generates elastin-derived peptides that contribute to the development of many age-related vascular pathologies. One of the main research topics of the UMR URCA/CNRS 7369 MEDyC concerns the elastin receptor complex, located in the plasma membrane and composed of three subunits: a receptor named Elastin Binding Protein, a protective protein, cathepsin A and a neuraminidase or sialidase, hNEU1. The hNEU1 is an exo-glycosidase that hydrolyzes a terminal sialic acid on glycoconjugates. This enzyme is a key regulator of the activation of a large number of membrane receptors responsible for various biological processes such as cell proliferation, cell signaling, inflammation or the development of cardiovascular diseases related to aging tissues. The development of inhibitors of human neuraminidase/sialidase hNEU1, a priority research topic for the ICMR's "Glycoscience and Modeling" (GeM) group in the framework of the 2018-2022 five-year project, would allow significant advances in the understanding of the development of age-related vascular diseases.
Most of the known neuraminidase inhibitors are not very selective, thus preventing their therapeutic use. Modeling and quantum docking studies on a model of NEU1 (Pr. E. Hénon, Dr. H. Khartabil, GeM group), carried out with the help of the Regional Computing Center ROMEO, have allowed us to select, in advance of this project, several molecular structures presenting, in silico, a good affinity for the active site of the enzyme. Among these target compounds, our research team has focused its attention on unsaturated sulfur analogues of a known inhibitor (DANA), analogues featuring moreover the possibility to introduce molecular diversity on the N-acyl group.
In the framework of an ICMR/MEDyC partnership, the objective of the proposed project is to design and synthesize new selective inhibitors of the hNEU1 sialidase as well as the biological evaluation of the prepared molecules.
A way to access this family of molecules, using as raw material D-xylose, a pentose abundantly present in lignocellulosic biomass, is proposed by a synthetic sequence using the skills of our team in the field of the synthesis of sulfur analogues of sugars.
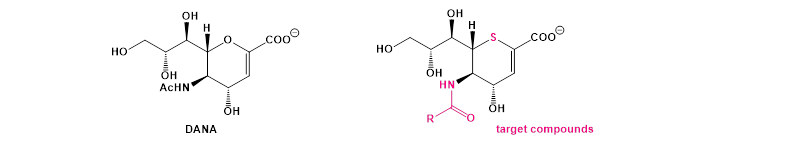
After the synthesis of the compounds, the biological and biochemical evaluation of the prepared target molecules will be carried out by measuring their capacity to inhibit the sialidase activity of NEU1 in two different cellular models.